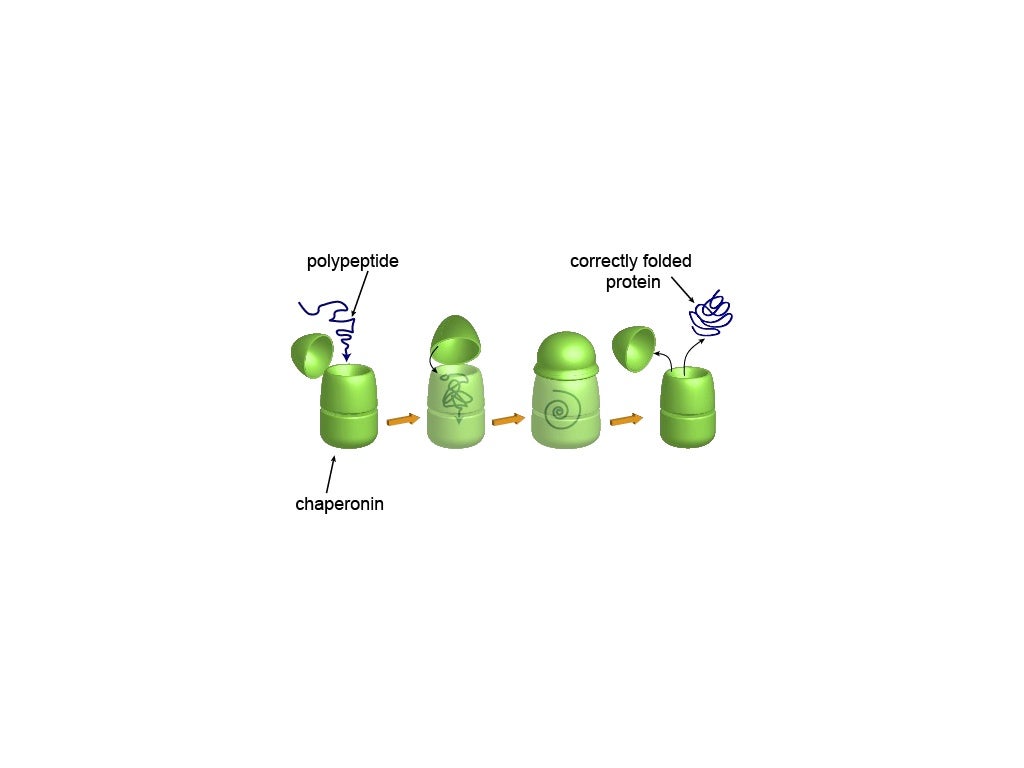
Given the central role of protein folding in biology, it is interesting to think that a misfolding can lead to dysfunction, modifying the cellular mechanism in which the protein is involved. Loss of protein structure can arise from alterations in diverse stages during protein synthesis, the degradation process, or changes in concentration of metabolites in the cell environment. Transmission and management of information through proteins is crucial for the homeostasis of the organisms, regulating mechanisms such as cell cycle, apoptosis, and cell growth. Most of the signaling proteins used by eukaryotic cells to communicate with their environment are assembled in endoplasmic reticulum (ER). Then, the understanding of the quality control checkpoints, which are performed during normal cell physiology, and those that are activated during protein stress, including the stress responses, function of catabolic machineries, and systems of communication among molecules and organelles, are subject to an extensive research. Proteomes have been described as multifaceted and constantly evolving entities. Likewise, cells must not only promote accurate protein folding but also prevent the accumulation of misfolded species that may arise from translation errors, and synthesis of aberrants mRNAs. Ĭells modulate protein folding and protein degradation through extensive signaling networks to avoid misfolded protein accumulation. Cytotoxic effects could be triggered by interactions among hydrophobic regions of proteins exposed to aqueous microenvironment with cellular biomolecules, such as other proteins, nucleic acids, or lipid membranes. 
For instance, oligomerization preceding amyloid fibril formation has been associated with cytotoxic effects, which may arise from their misfolded conformation in which hydrophobic side chains are exposed to the cytosol. This has allowed evolution to develop a conserved warning system, in which the exposure of protein hydrophobic regions is recognized as a molecular pattern associated with the presence of cytotoxicity. In this case, the structural stability of proteins depends largely on hydrophobic residues being oriented towards the protein core. Notwithstanding, although the process is extremely efficient, there is always the possibility that this accurate mechanism fails, and, in consequence, finding a protein folded into a non-native state becomes a reality. Cells have developed highly controlled mechanisms to maintain native protein folding, which include detail three-dimensional structure patterns and specific disordered domains. įunction in proteins largely depends on the acquisition of specific three-dimensional structures by folding at physiological time scales. For instance, establishment of preformed enzyme complexes for cell function has been proposed as a mechanism used by cells to rapidly respond to homeostasis unbalances, whereas pathways that regulate the conservation of protein folding have a key role. One of the keystones that has been considered to drive the evolution of organisms, relies on the capacity to detect, respond, and adapt to various stressors in the environment through cellular defensive mechanisms that protect the entire organism and maintain its capacity to grow and reproduce. This review centers on our current knowledge about pathways that modulate protein folding, and cell responses involved in protein homeostasis. Several diseases are known to have a molecular etiology in the malfunction of mechanisms responsible for protein folding and in the shielding of native structure, phenomena which ultimately lead to misfolded protein accumulation. We provide an overview of chaperone protein function, UPR via, spatial compartmentalization of protein folding, proteasome role, autophagy, as well as the intertwining between these processes. Therefore, taking this into consideration, a key part of this paper is developed around the protein folding phenomenon, and cellular mechanisms which support this pivotal condition. For instance, imbalances in secretory protein synthesis pathways lead to a condition known as endoplasmic reticulum (ER) stress which elicits the adaptive unfolded protein response (UPR). 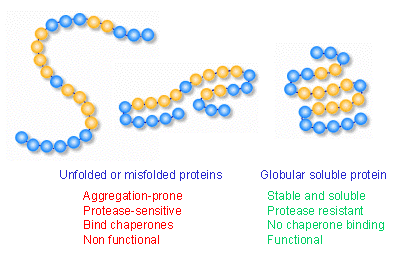
Interconnections among metabolic and cell signaling pathways are critical for homeostasis to converge on mechanisms associated with protein folding as well as for the preservation of the native structure of proteins. Therefore in recent years the etiology of a great number of diseases has been attributed to failures in mechanisms that modulate protein structure. Perturbations in these mechanisms can lead to protein dysfunction as well as deleterious cell processes. Highly sophisticated mechanisms that modulate protein structure and function, which involve synthesis and degradation, have evolved to maintain cellular homeostasis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
